
ParaSonic
A world without lice
67% of funding target
Highlights
Highlights
An innovative and effective solution to an age-old problem
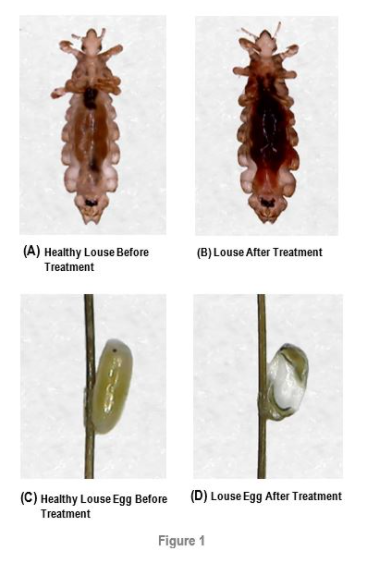
PanaSonic offers the first and only solution of its kind to the lice epidemic, the second largest health epidemic in the world of child care, in the form of the first ultrasonic comb, characterized by unprecedented efficiency alongside safety, in a quick and easy treatment. The ParaSonic comb is the only solution for the phenomenon of Super-Lice, lice that have developed complete resistance to existing treatments.

Over 94% efficacy proven in a successful clinical trial
ParaSonic's technology is ready for commercial production and marketing after it has been proven to be effective and safe, in a clinical research conducted at the Herzliya Medical Center during 2017-2018. 23 participants were treated and the procedure was found to be safe and effective, with no adverse effects reported, and over 94% of the lice eradicated. An additional clinical trial among 30 children is set to begin soon, in order to show the improvements achieved due to the product's upgrade regarding efficiency and battery use time. Mid-results of this new trial are expected by August 21.
Investing in an FDA-approved technology
ParaSonic has finalized all procedures requested by the FDA and has just received its approval for sales and marketing of the company’s product. During the corona period, the company has upgraded its product’s design and energy usage, in order to reach maximal efficiency in destroying lice and lice eggs. The clinical trial dedicated to evaluating these parameters is expected to increase the tech’s attractiveness, especially in this market which is waiting many years for innovation. The company is set to become a target for purchase by retail, medical and big pharma leaders, thanks to the great value it’s tech can generate for them.

A huge global market valued at $ billions annually
ParaSonic operates in the global market for lice and lice infestation products, estimated at billions of dollars annually, with sales of chemical products, prevention products, oils and combs totaling hundreds of millions annually in the US alone, as well as in Western Europe. So far, this market has not yet achieved a technological breakthrough, so the ParaSonic comb has no direct competitors, which is expected to help it retain a particularly significant market share.
Registered patents worldwide, attractive technological IP
The company’s product and technologies are protected by 2 "families" of patents, in registration procedures worldwide. These patents cover various facets, including application of the technology in the comb itself, the ultrasonic method of operation on which the product is based, and auxiliary accessories. The first patent series has already been approved in the US, Canada, Australia, and in the upcoming year the company will complete its registration also in Europe, China and Israel.

Ready for market, existing sales channels in the US and Israel
While finalizing the aforementioned registration processes, the company was recently granted sales approval for the US, and has already signed a vast commercial agreement to collaborate with the largest chain of clinics in the US for the treatment of lice, which includes hundreds of branches. ParaSonic is also in talks with another US partner, for an ongoing agreement which includes commitment to minimal orders of units per year. The company has signed an agreement with an Israeli distributor for direct online marketing in Israel, which will be used as a beta site, and as soon as next February the company will be able to hold an extensive survey among Israeli consumers.

Join a leading team that has already raised millions and performed exits
The company's team has extensive development and management experience and is led by CEO Mor Cohen. The current round is led by Avi Sharir, founder and CEO of Orad, an Israeli company purchased by Avid for millions of dollars, an investor with proven track record in the tech world, who was part of additional "exits", and has also officially joined the company's board.
The company's tech has also drawn media attention, among others in a Channel 2 News item, and the "Haolam Haboker'' news morning show. This is a chance to join a well-established company, after R&D and POC stages, which significantly disrupts its target market, and has already raised funds from private investors, the NGT technological greenhouse, as well as from the Chief Scientist’s program.

Pitch
Pitch
The Need
Lice is the second largest health issue in the world of children's medical care, after the flu epidemic. The problem is so common that it actually turned into a legitimate subject for children's books, such as "The Lice Nehama", a well-known children's book in Israel. However, despite the prevalence of the problem, there has been no technological leap forward in dealing with this problem to this day.
Lice infection occurs mainly in children aged 3-11. In the United States alone, 6-12 million new infections occur each year, with higher infection rates among girls, who usually have longer hair than boys.
For the economy, the lice epidemic means spending billions of $ - in total cost of OTC treatments, prescription treatments, mechanical combs, preventive treatments, and loss of work days for parents who need to stay home with their children.

A number of key factors reinforce the existing need:
1. Resistance - Lice are more resistant to the chemical treatments available today. In fact, every 2-3 years, lice become resistant to the new chemical products that are released to the market.
2. Efficiency against eggs - Even when the products are effective to some extent against the lice themselves, they are not effective enough against lice eggs. Therefore, treatment has to be repeated after several days to prevent further infection.
3. Side effects - The most common side effects to existing products on the market are skin irritation and allergies, resulting from the exposure of the scalp to these chemicals.
Due to possible side effects of drug lice products, and as a general trend in recent years, the market and consumers are increasingly choosing "natural" products or technological solutions. But the effectiveness of natural products today, made from different plant extracts, has not been clinically proven.
The solution
The lice epidemic refuses to disappear or diminish, and every year infection numbers rise. The phenomenon has been with us since the Stone Age – lice on the heads of human beings are actually modified chimpanzee lice, that has adapted themselves to man.
For a treatment more effective than ever before, ParaSonic developed an ultrasonic comb, which kills lice and lice eggs with a simple combing. In addition, the comb collects the dead head lice into a cartridge, so no rinsing is required after treatment.
Combing with ParaSonic comb enables treatment on a daily basis, which helps to prevent the spreading of lice and egg hatching on the child's head, as soon as they have been infected.
The Technology
ParaSonic's revolutionary comb is based on ultrasound technology and kills lice and eggs in a single combing session, taking about 10 minutes.
The teeth of the comb produce the ultrasound waves directed at the hair that passes between the teeth (this process is protected by the patent), so the lice and eggs on the hair are exposed to these waves and destroyed. The ultrasound waves are gentle pressure waves that operate at a specific frequency, designed to damage only the head and body lice (this frequency range is also protected by a patent).
CarelyTM is a comb with slightly elastic teeth, so the product is suitable for any hair type - smooth, curly or wavy - as well as for any hair thickness.
In the clinical trial conducted among 23 children at the Herzliya Medical Center, the kids reported that the combing was pleasant and expressed a preference for daily combing with CarelyTM, over a weekly combing with the simple, "traditional" comb.
How Does It Work?
Ultrasonic waves pass between the teeth of the comb. The lice and eggs that are found on the hair enter the acoustic area and are exposed to it. Only a second of exposure is needed in order for them to die.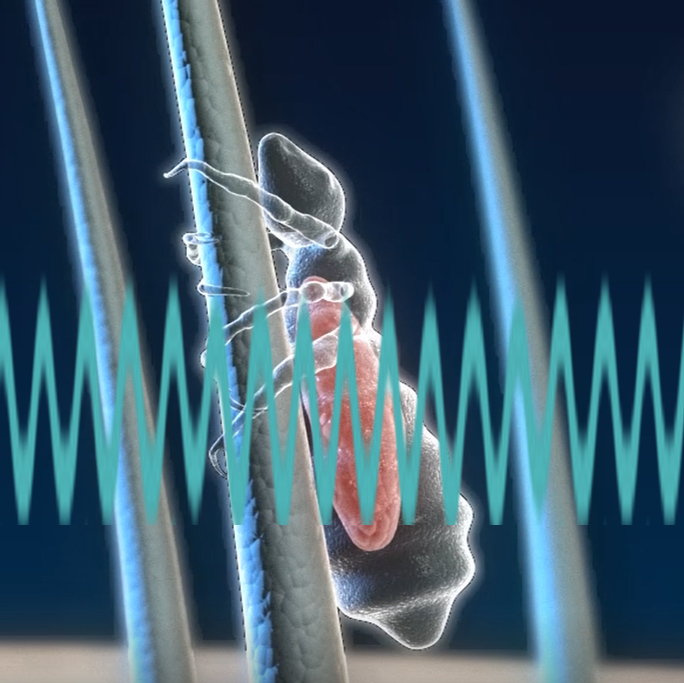
Team
Team
|
Mor has served as clinical director and product manager at leading Israeli medical esthetics companies. She holds an MSc in Biology/Genetics from Ben-Gurion University, and an MBA from Tel Aviv University.
|
|
CEO and Managing partner of NGT3VC 3, former CEO & director of Beta-O2 Technologies for 8 years. Served as CEO of the Technion Entrepreneurial Incubator for 11 years, led the establishment and investment in more than 50 companies including: Prolor Biotech (sold to OPKO, NYSE: OPK), Mazor Robotics (NASDAQ: MZOR), ReWalk (NASDAQ: RWLK), Corindus (NASDAQ: CVRS), Regentis and many others
|
|
Founder of 3D Innotech, with a vast technological and entrepreneurial background. Avi has held senior positions in a several technology groups, such as the Tadiran Communications Group and Orbotech. In 1993, Avi founded Ord High-Tech Systems, a leading provider of 3D real-time broadcast solutions. Avi already took part in two exits, including Ord High-Tech Systems Ltd.
|
|
Joe Kalfa is in the Dental Manufacturing and Distribution business, based out of Toronto, Canada.
He is an investor in a number of Dental & Med Tech start ups.
He also is a majority shareholder in a Luxury Consumer goods company based in the USA, that distributes Optical and other lines to companies like Walmart.
|



