ZygoFix
ZygoFix‘s zLOCK is a simple, elegant 3D-printed spinal fusion implant, which makes it the least invasive option for one of the most common spine surgeries
114% of funding target
Highlights
Highlights
zLOCK Takes Spinal Fusion to the 21st Century
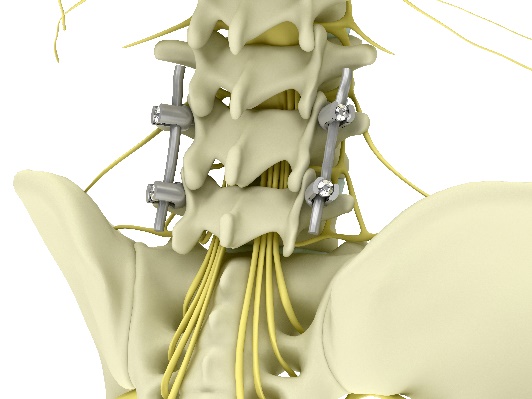
We all know the agony of a bad back. For half a million Americans being operated on every year due to chronic back pain such as ruptured discs, spinal fusion is the surgical solution. This huge Billions of Dollars market still utilizes 30-year-old technology of screws and rods that are invasive, complex to place, and may result in injury if misplaced.
 |
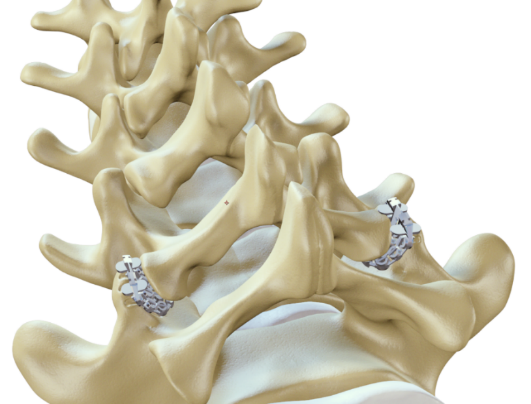
ZygoFix has developed a revolutionary screw-less implant for spinal fusion. Our technology uses advanced 3D printing to create a simple and smart implant. This means a much simpler and less invasive procedure, suitable for an outpatient setting.
 |
Let’s bring spinal fusion to the 21st century.
Minimizing and Simplifying Spinal Fusion
Today's standard method for spinal stabilization, is to create an external artificial metal bridge that holds the injured vertebrae together. This requires the surgeon to precisely drill 4 screws into the vertebrae and connect them with metal rods. ZygoFix's novel solution utilizes the natural bone structure of our vertebrae in order to provide the required stabilization. Instead of creating an artificial bridge, our unique 3D-printed implant is easily inserted and stabilizes the motion in its pivot point (the facet joint).
 |
 |
Feedback from Clinical Studies
In a 1-year post-operative visit, our first patient reported a great reduction in pain (on the VAS scale with 10 the most painful, patient reported 9/10 pre-operative to 1/10 post op. and 1/10 in 1-year post-operative).
”Patient doing extremely well and reports no pain! I can hardly believe it... I could discharge the patient even today! Practically no blood loss.” Email from surgeon 1-day post operation of first patient.
The clinical study is ongoing in the EU and in process to open an additional site in Israel.
Strong Market for Mergers and Acquisitions
The $9B spine market has been growing strong in M&A activities with larger companies buying innovative technologies that can strengthen their product offering.
Over 35 acquisitions were performed in the market during 2018, continuing into 2019 with a strong start and several substantial agreements.
ZygoFix's innovative product offers product diversity and a minimally invasive solution that can complement most any strategic player's portfolio.
Developed by leading innovators from the industry
ZygoFix benefits decades of knowledge in spine, and in medical device development and commercialization.
Founded by Uri Arnin and Prof. Yizhar Floman, two experienced entrepreneurs in spine technology.
Uri has been an inventor for 20+ years with over 40 patents in spine, and successfully started 3 companies in this field.
Prof. Floman is the Chairman and Director of the Israel Spine Center, Assuta Hospital, Tel Aviv; former Chief of Spine Surgery, Hadassah Hebrew University Medical Center, Jerusalem, President of the Israel Orthopedic Association
The company is supported by Trendlines Medical, Israel's largest medical seed investor and by the Israel Innovation Authority.
Pitch
Pitch
The problem
We all want to live life to the fullest. Chronic back pain is a leading limiting factor that may have substantial negative influence on our quality of life. For these people, spinal fusion surgery may be a life changer and is often recommended by their physician.
 |
The purpose of the procedure is to relieve the pressure applied on the nerves and stabilize that area of the spine. The standard procedure today is to drill 4 long screws into the spine and connect them together using rigid rods to create an artificial bridge between vertebrae.
These screws (called pedicle screws) are invasive – requiring 6 incisions, complex to place and may result in injury when misplaced.
6% of patient with fusion surgeries undergo a revision surgery due to complications. 50% of these complications are associated with the implants used such as a misplaced screw.
The market seeks a simpler and less invasive methods to stabilize the spine for fusion.
 |
The solution
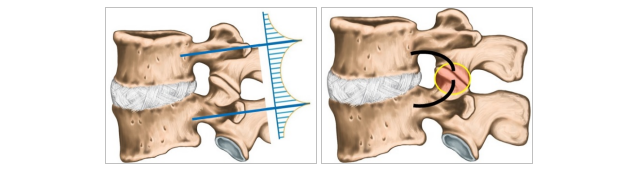
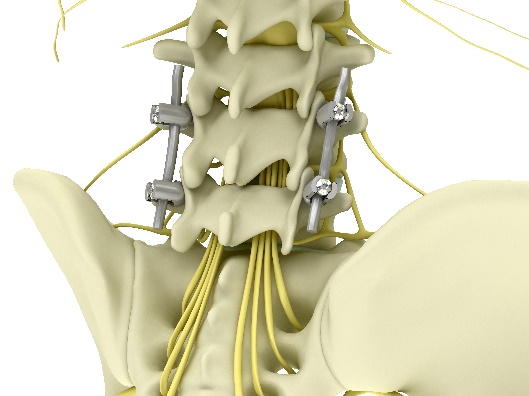
While surgeons are creating an artificial screw bridge, most of that bridge exists naturally in our spine's anatomy. Our vertebrae are connected by 2 small posterior joints (called facet joints). If we can lock these joints' motion, we essentially create the same bridge, just far less invasive and complex.
Left - Illustration of the external bridge built with screws and rods; Right – the natural bridge and facet joints circled
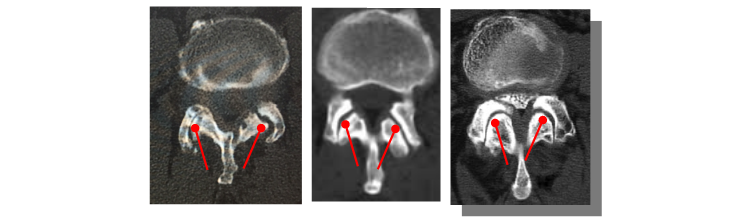
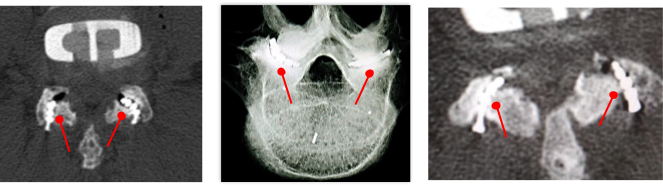
Now the challenge. Facet joints, like other joints, are not straight. They include a curve, which is different in every instance. So a rigid implant cannot be suitable.
Several examples of facet joint curves (in CT scans)
This challenge was the drive behinde ZygoFix's innovation. The zLOCK implant is the first implant to have the ability to shape itself automatically according to the joint's anatomy. The implant is designed as a combination of rigid and flexible elements, providing the ability to withstand the loads applied on the facet joint while also being capable to fit itself to the specific joint anatomy it is being inserted into. Keels located on both sides of the implant, anchor into the joint bones and immobilize it.
The zLOCK Implant
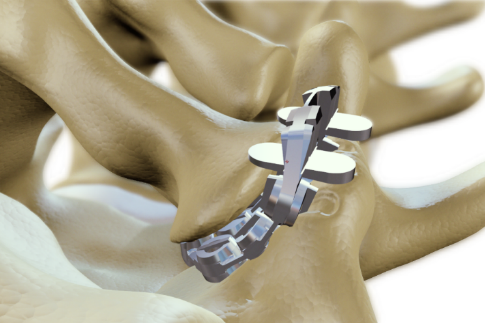 |
 |
Ability of implanted zLOCKs to take shape of facet different facet joins
The system includes a set of dedicated tools to perform the surgical procedure with a percutaneous approach (via a small incision in the skin).
This patented technology can also be utilized in other joint fusion applications.
zLOCK's Advantages
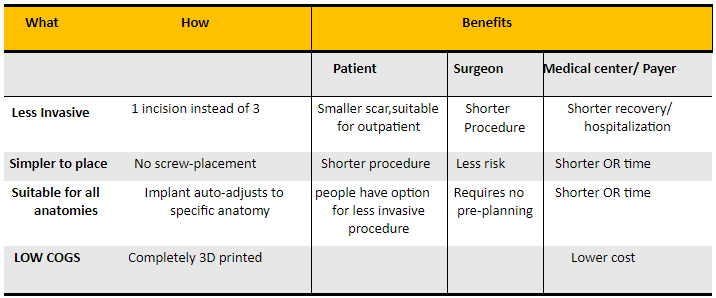
zLOCK's design provides several advantages beneficial to all:
- It is less invasive – a pedicle screw construct requires 2-3 incisions per side when placed in minimally invasive setting. zLOCK requires only 1 incision per side.
- The procedure is quick and simple – eliminating the complexity and risks of screw placement and its connection.
- As the implant customizes itself automatically, its placement is simple and straightforward.
- And, due to the fact that it is completely 3D printed, we maintain low and attractive COGS.
So it’s a smaller incision for the patient, a simpler and shorter procedure for the surgeon, and shorter usage of the OR (beneficial to hospitals, payers). Low COGS allows for higher margins and will also drive competitive pricing that benefits the customers.
IP Protection
ZygoFix's Intellectual Property is protected by 3 submitted patents. The first patent protects the implant design and is granted in the EU and under review in USA, Japan, China and Brazil. The other patents are in PCT stage and provide wider protection for similar designs and the used surgical tool.
 Big Potential
Big Potential
An average pedicle screw construct (4 screws and 2 rods) used to stabilize one segment or a vertebral pair costs $3,500 in the US.
The zLOCK implant is inline with market trends to minimize invasiveness in spine and in general surgery. zLOCK allows to take spine surgery a step forward by minimizing it and providing an alternative to the screw bridge (pedicle screw construct).
Achievements and Recognition
Our zLOCK system is outstanding and different in the spine market. We have received several recognitions to support this:
1. Spine Technology Awards – awarded in 2017 for best minimal invasive technology.
 |
2. Horizon 2020 seal of excellence – the prestigious European grant program has awarded ZygoFix with a seal of excellence for zLOCK along with winning PH-1 grant of €50,000.
 |
3. Market traction – 50+ inquiries from surgeons and distributors interested in use/ distribution from 18 different countries, 20+ from US.
An Experienced Team
Ofer Levy, CEO – 15+ years' experience in medical device industry; deep understanding of medical device development and lifecycle; Former Vice President of Qsite Regulatory and Quality; MBA Tel Aviv University, BSc mechanical engineering Ben-Gurion University.
Uri Arnin, Co-founder, CTO - Serial entrepreneur; 20+ years’ experience in medical device industry; 29 issued patents and 39+ pending (all in spine); formerly CTO Impliant (world’s first facet replacement device), founder and CTO ApiFix (MIS scoliosis correction system).
Prof Yizhar Floman, Co-founder, Medical Director - Chairman and Director of the Israel Spine Center, Assuta Hospital, Tel Aviv; former Chief of Spine Surgery, Hadassah Hebrew University Medical Center, Jerusalem; President of the Israel Orthopedic Association.
Our board is led by the chairman and CEO of the Trendlines Group, Todd Dollinger, who has led several exits in medical device.
And our advisory board includes experienced surgeons and business executives that are well connected to the US market and guide us toward it.

Team
Team
|
Serial entrepreneur in the field of spinal implants; 30 years’ experience in medical device industry; 29 issued patents and 43 pending (all in spine); former CTO, Impliant (world’s first facet replacement device); CTO, Spine 21 (world’s first remote-controlled spinal implant); founder and CTO, ApiFix??.
|
|
Chairman and Director, Israel Spine Center, Assuta Hospital, Tel Aviv; former Chief of Spine Surgery, Hadassah Hebrew University Medical Center, Jerusalem; President, Israel Orthopedic Association; member of numerous organizations, including Scoliosis Research Society, International Society for the Study of the Lumbar Spine, Spine Society of Europe, and the North American Spine Society.
|
|
Todd Dollinger brings decades of medical entrepreneurial experience to ZygoFix. Since 1991, Todd has been intricately involved with medical device start-ups bringing them form ideation to commercialization, IPOs and exits.
Todd is the Chairman and CEO of the Trendlines Group.
|
|
Barak Singer has over 15 years of experience in management, business development, investment banking, and venture capital.
Among others, Barak served as VP Business Development at Can-Fite BioPharma (NYSE: CANF) and CEO of its subsidiary Ophthalix, VP Business Development at Xenia Venture Capital, and was a Co-Founder and CEO at Or Capital Healthcare Partners.
|
|
Dr. Barcohana is a practicing spine surgeon and partner in Southern California Orthopedic Institute, LA, CA. He specializes in the surgical and nonsurgical treatment of spinal disorders involving the neck and back, utilizing microsurgical and minimally invasive techniques.
Dr. Barcohana attended UCLA and graduated Phi Beta Kappa, Summa Cum Laude, in neuroscience. He earned his medical degree from the University of California at San Francisco and performed his residency in orthopedic surgery in Stanford Univeristy.
|
|
Dr. Lewis is a board certified Neurosurgeon and the president of the Jackson Neurosurgery Clinic.
Dr. Lewis has completed his medical studies in Georgetown University School of Medicine with postgraduate studies in Neurosurgery in the University of Cincinnati College of Medicine.
|
|
Dr. Ira Fedder is a practicing orthopedic surgeon in Towson Orthopaedic Associates, University of Maryland, St. Joseph Medical Center.
He has received his Doctor of Pharmacy and M.D. degrees from the University of Maryland. His post graduate training was completed at University Hospital and Saint Joseph Medical Center. Dr. Fedder is fellowship trained in spine surgery.
|
|
Over 20 years experience in the US medical device industry. Successful commercialization and revenue generation at Zimmer Spine, Centinel Spine, Wenzel Spine and most recently Prosidyan which exclusive rights were acquired by DePuy Synthes in June, 2018. Mr. Luedke is currently, President & CEO of Spineway USA, Inc., a French based publicly traded company that treats severe spinal column pathologies.
|